Among 115 elements, metal and Non-metals are mainly two groups. Metals are 93 and non-metals 22. CBSE Class 10 with NCERT Solution.
Property of Metals:
Physical Properties of Metals:
(1) Physical State: Generally metals are hard at room temperature, example: iron, aluminium, gold etc. But mercury liquid at room temperature.
(2) Hardness: Generally most of the metals are hard and strong but some alkali metals like sodium, potassium are soft and can be cut by knife.
(3) Lusture: Generally metals have metallic lusture. They can be polished to give a highly reflective surface. But lead has dull appearance.
Property of Metals:
Physical Properties of Metals:
(1) Physical State: Generally metals are hard at room temperature, example: iron, aluminium, gold etc. But mercury liquid at room temperature.
(2) Hardness: Generally most of the metals are hard and strong but some alkali metals like sodium, potassium are soft and can be cut by knife.
(3) Lusture: Generally metals have metallic lusture. They can be polished to give a highly reflective surface. But lead has dull appearance.
(4) Sound: Metals produce sound when strike them, hence metal is called sonorous. For this property metals wire is used in musical instruments.
(5) Melting and boiling point: Generally metals have high melting and boiling point, except sodium, gallium, potassium.
(6) Density: Generally metals have high density except sodium, potassium.
(7) Ductility: By using force we can drawn metals in thin wires without breaking. Hence they are ductile. Except: zinc, mercury, gallium.
(8) Malleability: Metals are generally malleable means can be beaten into thin sheets without cracking.
(9) Brittleness: Metals are not brittle except zinc.
(10) Conduction: Metals generally are good conductor of heat and electricity. For this reason copper is used as electric wire.
(11) Solubility: Generally, metals do not dissolve in liquid solvents.
Chemical Properties of Metals:
(1) Number of valence Electrons: There are generally 1 or 2 or 3 electrons present in outermost shell. If there is more less number of valence electron, the metal is more active.
(2) Formation of ions: they form positive ions by losing one or more electrons from their valence shell.
(5) Melting and boiling point: Generally metals have high melting and boiling point, except sodium, gallium, potassium.
(6) Density: Generally metals have high density except sodium, potassium.
(7) Ductility: By using force we can drawn metals in thin wires without breaking. Hence they are ductile. Except: zinc, mercury, gallium.
(8) Malleability: Metals are generally malleable means can be beaten into thin sheets without cracking.
(9) Brittleness: Metals are not brittle except zinc.
(10) Conduction: Metals generally are good conductor of heat and electricity. For this reason copper is used as electric wire.
(11) Solubility: Generally, metals do not dissolve in liquid solvents.
Chemical Properties of Metals:
(1) Number of valence Electrons: There are generally 1 or 2 or 3 electrons present in outermost shell. If there is more less number of valence electron, the metal is more active.
(2) Formation of ions: they form positive ions by losing one or more electrons from their valence shell.
(3) Reaction with Oxygen: they form metal oxide when reacts with oxygen.
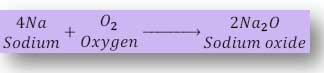
(i) When potassium reacts with oxygen forms potassium oxide and when sodium reacts with oxygen form sodium oxide. These reactions are take place rapidly since sodium, potassium are alkali metals.
(i) When potassium reacts with oxygen forms potassium oxide and when sodium reacts with oxygen form sodium oxide. These reactions are take place rapidly since sodium, potassium are alkali metals.
(ii) When magnesium reacts with oxygen, it burns with dazzling flames and produce magnesium oxide.
(iii) Normally aluminium does not react with oxygen but when it burns in air produce aluminium oxide. Similarly In case of zinc and iron which do not react with oxygen but when strongly heated formed zinc oxide and iron oxide respectively.
Reaction with water:
All the metals do not reacts with metal but alkali metals like sodium, potassium etc reacts with water and produce hydrogen gas. When metal reacts with water produce hydrogen gas and their hydroxide.
Metal + Water → Hydrogen gas + Metal Hydroxide
(i) Sodium or potassium vigorously reacts with water produce hydrogen gas and evolve a large amount of heat during this reaction.
All the metals do not reacts with metal but alkali metals like sodium, potassium etc reacts with water and produce hydrogen gas. When metal reacts with water produce hydrogen gas and their hydroxide.
Metal + Water → Hydrogen gas + Metal Hydroxide
(i) Sodium or potassium vigorously reacts with water produce hydrogen gas and evolve a large amount of heat during this reaction.
(ii) Calcium also reacts fairly vigorously with cold water and form hydrogen gas and calcium hydroxide.
(iii) The reaction between magnesium and water take place very slowly and forms hydrogen gas and magnesium oxide.
(iv) The strongly heated aluminium, zinc, iron reacts with steam and forms hydrogen gas with aluminium oxide, zinc oxide, iron oxide respectively.












 RSS Feed
RSS Feed